Abstract
Background: Intracellular Nicotinamide Adenine Dinucleotide (NAD+) is a vital coenzyme that naturally declines with age, leading to mitochondrial dysfunction, metabolic issues, and reduced sleep quality. NMN (Nicotinamide Mononucleotide) serves as a direct NAD+ precursor within the salvage pathway, effectively restoring cellular health and energy metabolism. Strategies aimed at restoring NAD+ levels have the potential to impact both metabolic and neurobehavioral outcomes.
Objective: This exploratory single-arm study evaluated the changes in intracellular NAD+, metabolic and inflammatory biomarkers, and self-reported mood and sleep after 60 days of daily supplementation with AVEA NAD+ Booster and β-nicotinamide mononucleotide (AVEA NMN; 500mg).
Methods: In a 60-day intervention, 28 participants (mean age ≈ 52 years) consumed daily oral doses of the AVEA Booster (a synergistic blend of Trans-Resveratrol, Ubiquinol (CoQ10), Betaine HCl, Pterostilbene, Apigenin, and Nicotinamide) combined with AVEA NMN. Biomarkers (intracellular NAD+, insulin, hsCRP) and validated self-reported measures (DASS-21, WHO-5, MOS Sleep Scale) were assessed pre- and post-intervention. Paired statistical analyses were conducted with significance set at p < 0.05.
Results: The supplementation protocol was well tolerated. The preliminary findings indicated a significant increase in intracellular NAD+ (by 44%, p < 0.0001; Cohen’s d = 1.39), with directionally favorable but non-significant changes in insulin and hsCRP (decreased by 30%, p = 0.1254). Improvements in selected self-reported sleep measures and mood were seen, with depression scores decreasing by 38% (p = 0.0499), and sleep disturbance decreasing by 24% (p = 0.0080). Sleep Problems Index II improved by 15% (p = 0.0425).
Conclusion: Sixty days of AVEA NAD+ Booster and AVEA NMN combined supplementation significantly enhances intracellular NAD+ and provides measurable improvements in mood and sleep. For individuals seeking the best NMN supplement or a comprehensive NAD+ booster, this synergistic combination offers a scientifically-backed solution for healthy aging and vitality.
Keywords: NAD+ Booster, NMN Supplement, AVEA NMN, Nicotinamide Mononucleotide, Best NAD+ Supplement, Longevity Supplements, Trans-Resveratrol, Anti-Aging, Sleep Quality, Mood Support.
NAD longevity, L-tryptophan, salvage pathway, health supplements, inflammation, aging, energy, focus
In this study
Introduction
Aging is associated with a significant decline in intracellular Nicotinamide adenine dinucleotide (NAD+) levels, an essential coenzyme for cellular energy metabolism and a substrate for enzymes that regulate DNA repair, stress responses, circadian rhythms, and inflammation. This reduction is closely linked to the development of chronic cardiovascular, neurodegenerative, and metabolic diseases1. Declining NAD+ levels are a hallmark of aging, linked to chronic inflammation and metabolic dysfunction, which may contribute to fatigue, poor sleep, and reduced psychological well-being2,3. These observations have positioned NAD+ metabolism as a central therapeutic target in age-related disease and aging biology.
NMN as a NAD+ Precursor
NAD⁺ is synthesized via salvage pathways from vitamin B3 family precursors. β-nicotinamide mononucleotide (NMN) is a direct NAD+ precursor in the salvage pathway, and thereby supports sirtuin and PARP activity4, mitochondrial oxidative phosphorylation, and redox homeostasis5. Age-related upregulation of CD38 (a NAD⁺‑consuming enzyme) depletes NAD+ levels, disrupting the CD38/NAD+/SIRT1 axis and mitochondrial homeostasis6. Conceptually, boosting NAD⁺ precursors while limiting NAD⁺ degradation and supporting mitochondrial electron transport provides a combined strategy to restore cellular bioenergetics and stress resilience7,8.
As a direct NAD⁺ precursor, NMN effectively boosts blood and intracellular NAD⁺ levels in humans following oral administration at doses of 250–900 mg/day, with clear dose–response relationships in randomized, placebo‑controlled trials9 .
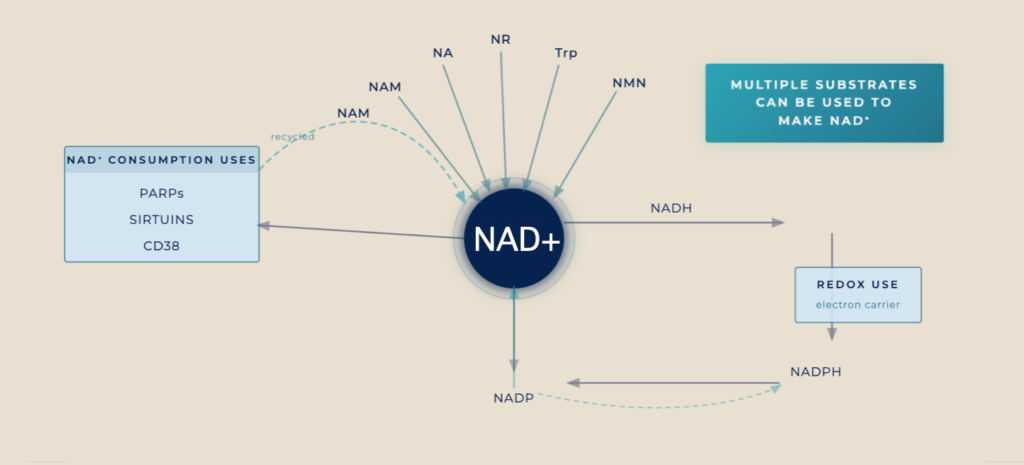
AVEA NAD+ Booster: Mechanistic Rationale and Evidence
| Ingredient | Primary role in NAD⁺ / mitochondrial axis | Expected clinical outcomes in Aging |
| Nicotinamide Adenine Dinucleotide Precursor NMN (500 mg). | Direct NAD⁺ precursor via salvage pathway; raises blood NAD⁺ and activates NAD⁺‑dependent signaling | Increases blood and intracellular NAD+ levels. Improved mitochondrial function, enhanced metabolic homeostasis, increased physical performance, and vascular health |
| Veri-te™ Trans-Resveratrol (150 mg) | Activates SIRT1/PGC‑1α, enhances mitochondrial biogenesis, antioxidant and vasoprotective effects | Supports mitochondrial biogenesis, reduces oxidative stress and inflammation, potentially improving cardiovascular health and longevity |
| Pterostilbene (50 mg) | Resveratrol analog with higher bioavailability; similar sirtuin/AMPK signaling | Enhanced antioxidant defenses, improved metabolic regulation, and anti-inflammatory effects |
| Kaneka Ubiquinol® [Coenzyme Q10] (100 mg) | Electron carrier in mitochondrial ETC; supports ATP generation and reduces oxidative stress | Improved cellular energy production, reduced oxidative damage, potential mitigation of age-related fatigue |
| Apigenin (50 mg) | Flavonoid with CD38‑inhibiting and NAD⁺‑sparing potential; anti‑inflammatory and antioxidant | Preservation of NAD⁺ levels, reduced chronic inflammation, improved mitochondrial function |
| Nicotinamide [Vitamin B3] (16 mg) | Vitamin B3; salvage pathway precursor (NAM → NMN → NAD⁺) | Supports NAD⁺ biosynthesis, may improve metabolic health and reduce age-associated decline |
| Betaine Hydrochloride (50 mg) | Methyl donor supporting homocysteine remethylation; indirect support of one‑carbon and redox metabolism | May lower homocysteine levels reducing cardiovascular risk; supports methylation reactions important for cellular health |
Despite extensive studies on the individual benefits of these components, no research to date has evaluated their combined effects in a single supplementation regimen. This pilot study aims to fill this gap, investigating the synergistic potential of these compounds to produce measurable changes in intracellular NAD+ levels, thus potentially influencing NAD+ metabolism, systemic inflammation, and overall health.
Results
Biomarkers
Following the intervention, several biomarkers showed changes. NAD+ increased by 44% (p < 0.0001; Figure 1) with a large effect size (Cohen’s d = 1.39). hsCRP decreased by 30%; however, this reduction was not statistically significant (p = 0.1254; Figure 1).
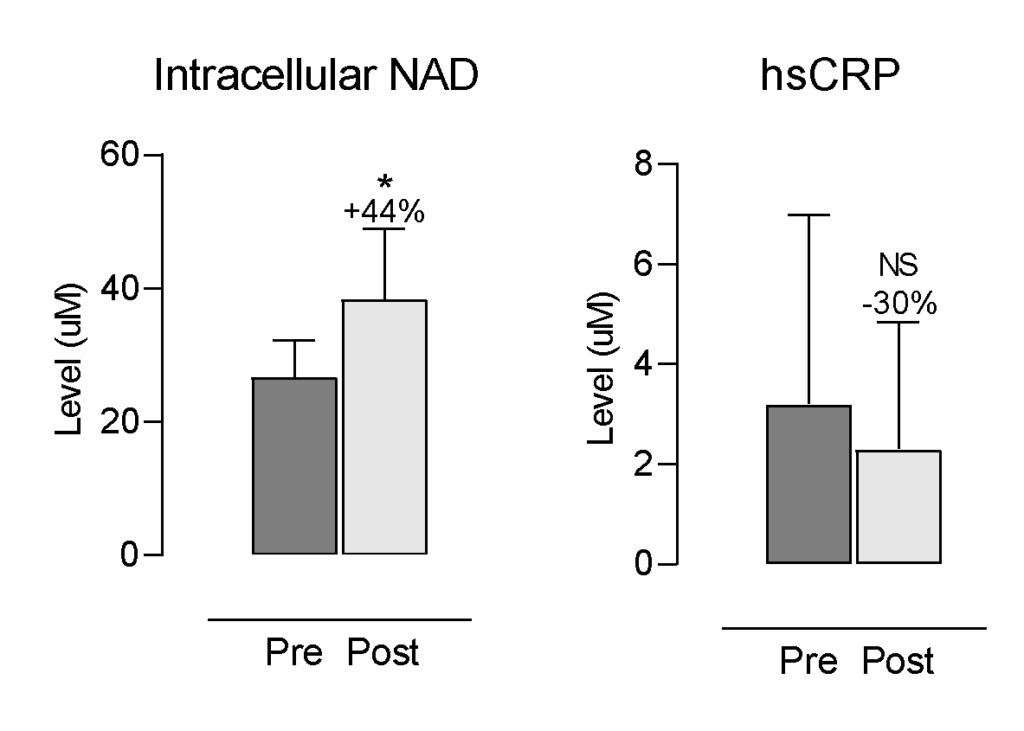
Figure 1. Changes in NAD+ and hsCRP Levels. Each bar represents the mean and standard deviation (SD) for 28 and 25 participants respectively. * indicates p < 0.05; NS = non-significant. Percent change = (post–pre)/pre×100.
A significant increase in NAD+ levels suggests enhanced metabolic activity. Reductions in insulin and hsCRP may indicate trends toward improved insulin sensitivity and lower inflammation, though not statistically significant.
Psychological Measures
The DASS-21 questionnaire evaluates self-reported levels of Depression, Anxiety, and Stress, with higher scores indicating more severe outcomes10. The DASS-21 questionnaire revealed a statistically significant reduction in self-reported depression scores, with a 38% decrease (p = 0.0499). Stress scores decreased by 37%, showing a trend toward significance (p = 0.0802), while anxiety scores remained unchanged (p = 0.8112; Figure 2).

Figure 2. Self-Reported Changes in DASS-21 Scores. Each bar represents the mean and standard deviation (SD) for 28 participants. * indicates p < 0.05; NS = non-significant. Percent change = (post–pre)/pre×100.
The WHO-5 has been developed to assess well-being11. In this questionnaire, a percentage score of zero represents the worst possible mental well-being; a score of 100 represents the best possible mental well-being. The WHO-5 Well-Being Index showed a 6% increase, which was not statistically significant (p = 0.2501; Figure 3).
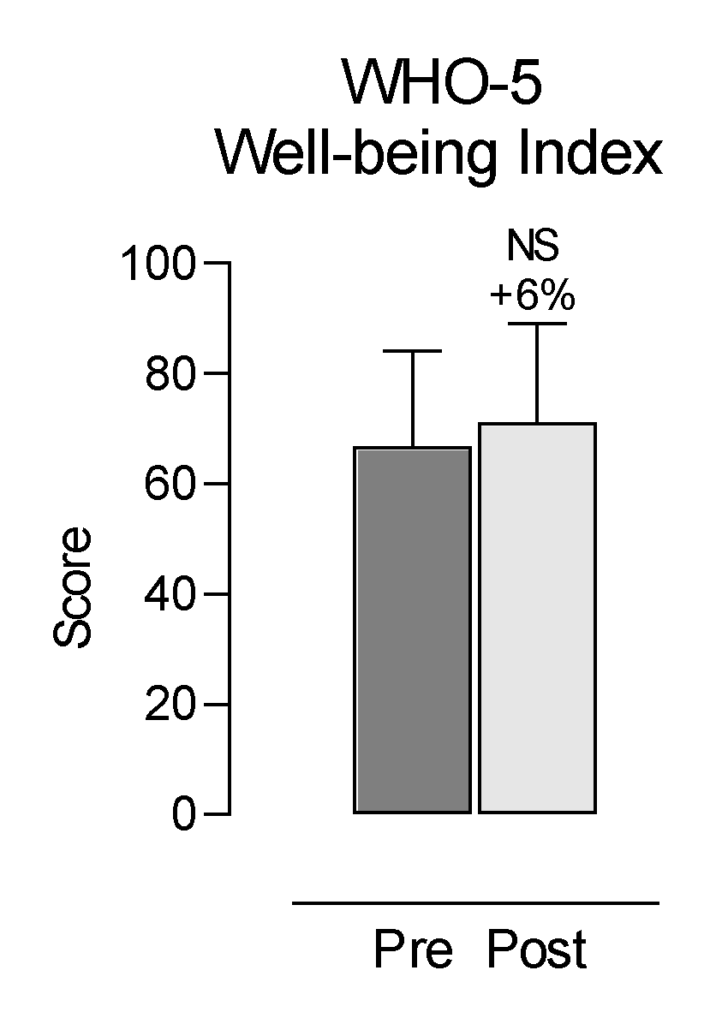
Figure 3. Self-Reported Changes in WHO-5 Well-Being Index. Each bar represents the mean and standard deviation (SD) for 28 participants. NS = non-significant. Percent change = (post–pre)/pre×100.
Depression scores significantly decreased, and stress showed a trend toward improvement, suggesting potential mood benefits.
Sleep Measures
The self-reported MOS Sleep Scale (Medical Outcomes Study Sleep Scale) evaluates key dimensions of sleep over the past four weeks through 12 items, grouped into seven subscales: sleep disturbance, snoring, shortness of breath/headache, sleep adequacy, somnolence, sleep problems index I, and sleep problems index II12. Responses are scored and transformed into a 0–100 scale, where higher scores reflect more of the concept being measured (e.g., more disturbance or better adequacy, depending on the subscale). Two additional measures—sleep quantity (hours of sleep) and optimal sleep (7–8 hours per night)—are also derived.
Participants reported improvements in several aspects of sleep. The most notable changes were a 24% reduction in sleep disturbance (p = 0.0080; Figure 4) and a 15% reduction in Sleep Problems Index II (p = 0.0425; Figure 4), both statistically significant. Sleep Problems Index I decreased by 13%, showing a trend towards significance (p = 0.0950; Figure 4).
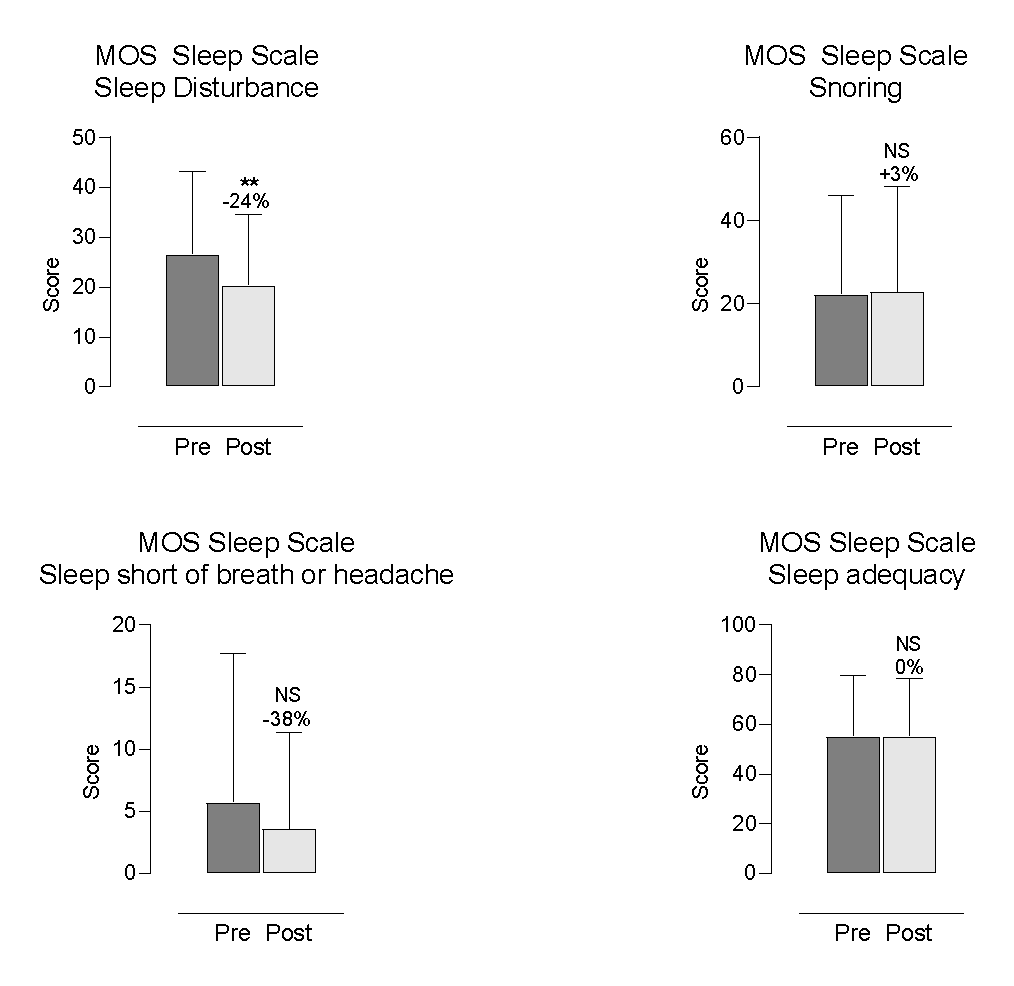
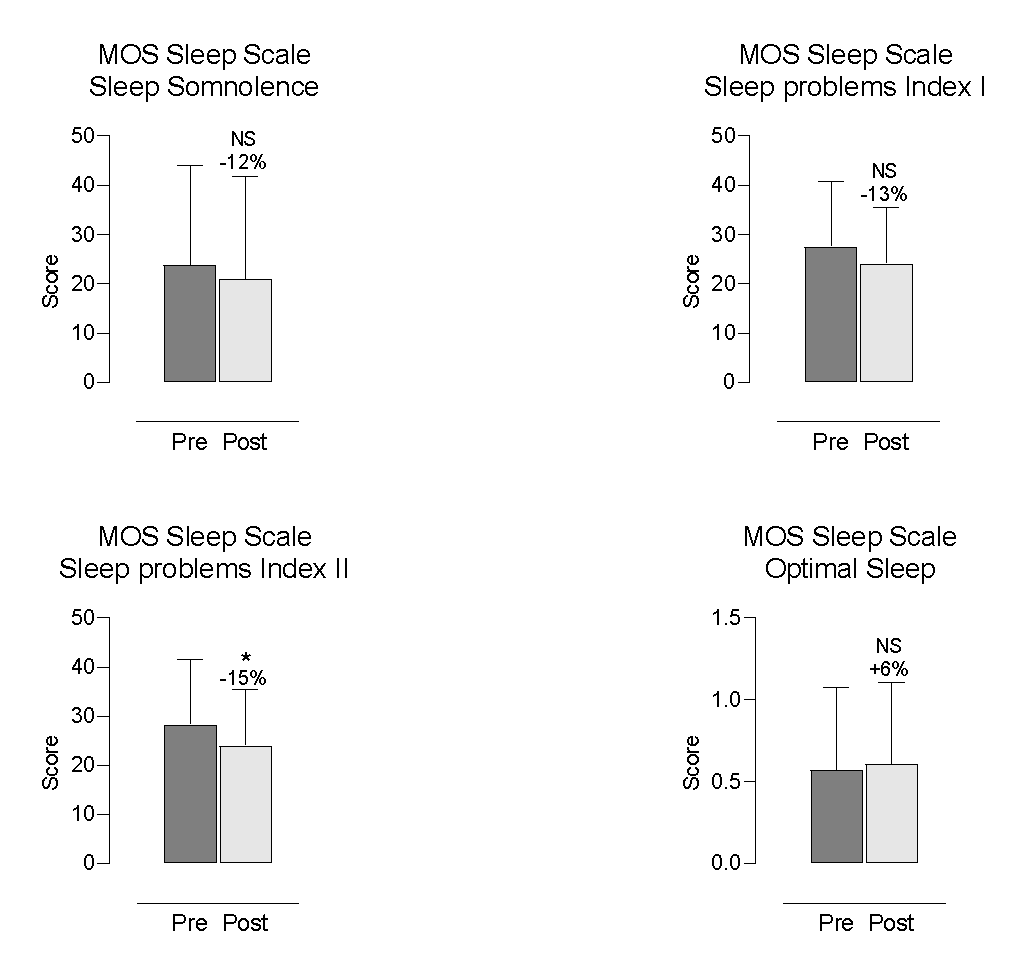

Figure 4. Self-Reported Changes in MOS Sleep Scale Outcomes
Each bar represents the mean and standard deviation (SD) for 28 participants. * indicates p < 0.05; NS = non-significant. Percent change = (post–pre)/pre×100.
Other self-reported sleep parameters—including snoring (+3%; p = 0.8490; Figure 5), shortness of breath or headache during sleep (-38%; p = 0.4375; Figure 5), sleep adequacy (0%; p > 0.9999; Figure 6), somnolence (-12%; p = 0.2982; Figure 6), and optimal sleep (+6%, mean = 0.61; p > 0.9999; Figure 6)—did not exhibit statistically significant changes.
Participants reported significantly better sleep, particularly reduced disturbance and fewer overall sleep problems. Other domains showed minor or non-significant changes.
Adverse Events Monitoring
Participants completed weekly monitoring forms to report any side effects or discomfort potentially associated with the supplementation protocol. The vast majority of participants (89%) reported no adverse events throughout the study period. A small number of participants (n = 3) noted mild symptoms, including dizziness, fatigue, and tiredness. One participant mentioned uncertainty about whether these effects were attributable to the supplements or other lifestyle factors such as alcohol or caffeine intake. These self-reported effects were non-serious, transient, and did not lead to participant withdrawal. Overall, the supplementation protocol appeared to be well-tolerated.
Discussion
The 60-day supplementation protocol using two AVEA products— AVEA NAD+ Booster and AVEA NMN—was associated with several promising wellness-related effects. Most notably, NAD+ levels increased by 44%, indicating a substantial enhancement in cellular energy metabolism. Although insulin and hsCRP reductions did not reach statistical significance, both trended downward, suggesting potential improvements in insulin sensitivity and inflammation that warrant further investigation
From a psychological perspective, participants reported meaningful benefits: self-reported depression scores decreased significantly, and stress scores showed a trend toward improvement, suggesting a potential mood-enhancing effect of the supplementation. Self-reported sleep quality also improved, with significant reductions in sleep disturbance and Sleep Problems Index II scores.
This study offers valuable preliminary insights, but has several limitations that must be considered when interpreting the results. The lack of a placebo or control group means that it is not possible to isolate the effects of the intervention from other factors such as time, lifestyle changes, or placebo effects. Therefore, no causal relationship between the supplements and the observed outcomes can be confirmed.
In addition, the small sample size of only 28 participants may be underpowered to detect smaller but potentially meaningful effects. Some non-significant trends may have reached significance in a larger sample.
Finally, the short duration of the intervention evaluated short-term responses only. It is unclear whether the observed changes—especially in biomarkers and psychological well-being—are sustained over time.
Conclusion
Overall, despite the aforementioned limitations, the findings point to potential wellness benefits of the AVEA supplementation protocol, particularly in terms of mood, sleep quality, and NAD+ enhancement. Several physiological trends are encouraging, but larger, placebo-controlled studies with longer follow-up are needed to confirm these preliminary observations and to better understand their clinical relevance.
Material and Methods
Study design
This study included a total of 28 participants: 18 females (mean age = 52.11) and 10 males (mean age = 52). Participants were enrolled based on the following inclusion criteria: adults aged 18–65 without major diagnosed conditions and not currently using new therapies or supplements, ability to provide informed consent, and willingness to comply with all study procedures.
The intervention consisted of a 60-day supplementation protocol using two AVEA products.
- Booster: containing Veri-te™ Trans-Resveratrol (150 mg), Kaneka Ubiquinol® [Coenzyme Q10] (100 mg), Betaine Hydrochloride (50 mg), Pterostilbene (50 mg), Apigenin (50 mg), and Nicotinamide [Vitamin B3] (16 mg).
- NMN: containing Nicotinamide Adenine Dinucleotide Precursor (500 mg).
Participants were instructed to take one capsule of each product daily by oral route, every morning with or without food. Data were collected before and after the intervention using a combination of:
- Biomarker measurements
Biomarkers, measured through blood samples, including NAD+, insulin, and hsCRP;
- Self-reported questionnaires
Self-reported questionnaires for psychological and sleep outcomes, including the DASS-21 (Depression, Anxiety, and Stress Scale), WHO-5 Well-Being Index, and Medical Outcomes Study (MOS) Sleep Scale.
Statistical analysis
All data were analyzed using GraphPad Prism 8.0. Prior to analysis, datasets were inspected for outliers, which were removed where appropriately justified. The Shapiro-Wilk test was conducted to assess the normality of each variable. Depending on distribution characteristics and study design, either paired or unpaired parametric t-tests or non-parametric equivalents were applied to compare values across time points. Descriptive analyses were also performed: for normally distributed data, results are reported as mean with 95% confidence intervals (CI); for non-normally distributed data, median with 95% CI is presented. Effect sizes were calculated to provide additional context on the magnitude of observed changes. A significance level of p < 0.05 was used throughout. P-values are nominal (unadjusted) given the exploratory design.
References
- Kolotyeva NA, Groshkov AA, Rozanova NA, et al. Pathobiochemistry of Aging and Neurodegeneration: Deregulation of NAD+ Metabolism in Brain Cells. Biomolecules. 2024;14(12):1556. doi:10.3390/biom14121556.
- Qin Song, Xiaofeng Zhou, Kexin Xu, Sishi Liu, Xinqiang Zhu, Jun Yang. The Safety and Antiaging Effects of Nicotinamide Mononucleotide in Human Clinical Trials: an Update. Advances in Nutrition 1416-1435 (2023)2023.
- Yu B, Jing X, Jia L, Wang M, Liu L, Ping S, Wang Y, Yang M. The versatile multi-functional substance NMN: its unique characteristics, metabolic properties, pharmacodynamic effects, clinical trials, and diverse applications. Front Pharmacol. 2024;15:1436597.
- Covarrubias AJ, Perrone R, Grozio A, Verdin E. NAD+ metabolism and its roles in cellular processes during ageing. Nat Rev Mol Cell Biol. 2021 Feb;22(2):119-141. doi: 10.1038/s41580-020-00313-x. Epub 2020 Dec 22. PMID: 33353981; PMCID: PMC7963035.
- Levine, D. C., Hong, H., Weidemann, B. J., Ramsey, K. M., Affinati, A. H., Schmidt, M. S., Cedernaes, J., Omura, C., Braun, R., Lee, C., Brenner, C., Peek, C. B., & Bass, J. (2020). NAD+ controls circadian reprogramming through PER2 nuclear translocation to counter aging. Molecular Cell, 78(5), 835-849.e7. https://doi.org/10.1016/j.molcel.2020.04.010
- Braidy, N., Berg, J., Clement, J., Khorshidi, F., Poljak, A., Jayasena, T., Grant, R., & Sachdev, P. (2019). Role of nicotinamide adenine dinucleotide and related precursors as therapeutic targets for age-related degenerative diseases: Rationale, biochemistry, pharmacokinetics, and outcomes. Antioxidants & Redox Signaling, 30(2), 251–294. https://doi.org/10.1089/ars.2017.7269
- Xie, N., Zhang, L., Gao, W. et al. NAD+ metabolism: pathophysiologic mechanisms and therapeutic potential. Sig Transduct Target Ther 5, 227 (2020). https://doi.org/10.1038/s41392-020-00311-7
- Katsyuba, E., Romani, M., Hofer, D., & Auwerx, J. (2020). NAD+ homeostasis in health and disease. Nature Metabolism, 2(1), 9–31. https://doi.org/10.1038/s42255-019-0161-5
- Katayoshi, T., Uehata, S., Nakashima, N., Nakajo, T., Kitajima, N., Kageyama, M., & Tsuji-Naito, K. (2023). Nicotinamide adenine dinucleotide metabolism and arterial stiffness after long-term nicotinamide mononucleotide supplementation: a randomized, double-blind, placebo-controlled trial. Scientific Reports, 13(1), 2786. https://doi.org/10.1038/s41598-023-29787-3
- Antony, M. M., Bieling, P. J., Cox, B. J., Enns, M. W., & Swinson, R. P. (1998). Psychometric properties of the 42-item and 21-item versions of the Depression Anxiety Stress Scales in clinical groups and a community sample. Psychological Assessment, 10(2), 176–181. https://doi.org/10.1037/1040-3590.10.2.176
- Kliem, S., Lohmann, A., Fischer, S., Baier, D., Clemens, V., Sachser, C., Kampling, H., Brähler, E., & Fegert, J. M. (2025). Psychometric evaluation and updated community norms of the WHO-5 well-being index, based on a representative German sample. Frontiers in Psychology, 16, 1592614. https://doi.org/10.3389/fpsyg.2025.1592614
Hays, R. D., Martin, S. A., Sesti, A. M., & Spritzer, K. L. (2005). Psychometric properties of the Medical Outcomes Study Sleep measure. Sleep Medicine, 6(1), 41–44. https://doi.org/10.1016/j.sleep.2004.07.006












