Abstract
Background: Aging leads to a decline in natural collagen synthesis, resulting in wrinkles, reduced skin elasticity, hydration, and structural integrity. While standard supplements vary in efficacy, The Avea Collagen Activator utilizes Colgevity™ (a patented, science backed amino acid ratio of 3:1:1 glycine, proline, hydroxyproline) has demonstrated preclinical and human translational relevance in collagen biology and longevity research.
Objective: This study evaluates the impact of a precursor-based collagen supplement on dermatological quality and epigenetic aging (DNA methylation) in healthy adults
Methodology: In a six-month observational human clinical study (N=58, age ≥35), participants consumed one daily sachet of Avea Collagen Activator. Assessments included Visia imaging for texture, Corneometer for hydration, Cutometer for elasticity, and TruMe labs saliva tests for biological age. Statistical analyses included repeated-measures testing and paired comparisons.
Results: Significant improvements were observed in skin texture (+8.5%), hydration (+34.6%), and skin elasticity (+5.0%) within three months. Mean biological age decreased by approximately 1.4 years after six months of supplementation (p=0.040). Individuals with higher baseline biological ages experienced the most dramatic results, with some “gaining back” up to 12 years.
Conclusion: Avea’s Collagen Activator is a premier, non-invasive solution for those seeking the best collagen supplement for anti-aging. By optimizing the specific amino acid precursors required for collagen synthesis, it provides measurable improvements in both visible skin quality and internal biological longevity.
Keywords: Collagen supplement, Avea Collagen Activator, Colgevity, reverse biological age, skin hydration, DNA methylation age, anti-aging amino acids, Calcium AKG, skin elasticity.
In this study
Background
Collagen is one of the most abundant proteins, making up 12-17% of the total proteins in mammals and forming the primary scaffold of skin, bone, cartilage, and tendons1-4. In the dermis, type I collagen provides mechanical strength, elasticity, and firmness5. As we age, the body’s natural ability to replenish collagen decreases by approximately 1.0%-1.5% per year 6, a central driver of visible skin ageing. With advancing age and cumulative exposure to intrinsic (genetic, hormonal) and extrinsic (UV radiation, pollution, smoking) factors, dermal collagen becomes fragmented and its overall content declines, contributing to dermal thinning, wrinkle formation, laxity, and impaired barrier function1,5,7.
Over the last few years, collagen supplementation has gained traction for health improvements. In 2024, the global collagen market size was estimated at around 9.4 billion USD, with a compound annual growth rate of 5.4%. It is projected to be roughly 16 billion USD by 20338. Oral collagen supplementation has emerged as a non‑invasive strategy to counteract these changes9. Multiple randomized controlled trials and meta‑analyses report that hydrolyzed collagen and collagen peptides improve skin hydration, elasticity, dermal density, and wrinkle severity after 6–12 weeks of intake, generally with a good safety profile10. These effects are often enhanced when collagen is combined with micronutrients and antioxidants (e.g., vitamin C, vitamin E, zinc, acerola), which support collagen biosynthesis and protect against oxidative damage 2,11.
Against this background, targeted amino acid formulations that mimic collagen’s characteristic composition, particularly glycine, proline, and hydroxyproline, are of special interest2. These residues dominate collagen peptides, are rapidly absorbed as bioactive di‑ and tripeptides, and can stimulate fibroblast activity, extracellular matrix synthesis, and skin barrier improvement12.
Avea’s Collagen Activator has been designed to support collagen synthesis using a patented blend of amino acids, 3 glycine – 1 proline – 1 hydroxyproline (Colgevity™), Calcium AKG, and antioxidants such as Astaxanthin and vitamin C (Table 1).
Table 1: Key Active Ingredients and Mechanistic Roles
| Ingredient | Primary mechanistic role in Skin and Collagen | Expected clinical outcomes in Aging |
| Glycine–Proline–Hydroxyproline (3:1:1, Colgevity™) | Provides core collagen amino acids; absorbed as bioactive di/tripeptides (e.g., Gly‑Pro‑Hyp); stimulates fibroblast activity, collagen and HA synthesis; may reduce MMP‑mediated degradation | Improved dermal collagen density, elasticity, hydration; reduced wrinkle depth and roughness after 6–12 weeks |
| Calcium α‑ketoglutarate | Central metabolite in TCA cycle; supports cellular energy and nitrogen metabolism; may influence ECM turnover and collagen homeostasis under stress | Potential support of dermal matrix maintenance and resilience during ageing |
| Astaxanthin | Potent antioxidant carotenoid; quenches ROS; protects lipids and proteins from photo‑oxidative damage | Reduced oxidative collagen damage; support for elasticity, firmness, and barrier integrity |
| Vitamin C | Essential cofactor for prolyl/lysyl hydroxylases in collagen biosynthesis; antioxidant; supports regeneration of other antioxidants | Enhanced collagen maturation and stability; improved dermal structure, hydration, and reduced wrinkles when combined with collagen peptides |
In this observational human study, we aimed at validating its efficacy in human subjects. Previous research has shown that the proper ratio of three glycine to one proline to one hydroxyproline was sufficient to increase the healthspan and lifespan of C. elegans, as well as collagen homeostasis in human fibroblasts in vitro. Here, we demonstrated that combining a specific ratio of collagen amino acids is not only safe in humans, but also contributed to improved skin quality within three months of intake. Additionally, It also reduced biological age by 1.4 years upon 6 months of oral supplementation.
Results & Discussion
Safety assessment and descriptive statistics of participants characteristics
We screened 102 participants and confirmed that 66 participants were eligible for the study (Fig. 1a). The dropout rate due to adverse events was 6%, with four out of 66 participants experiencing mild, transient side effects that resolved without intervention (Fig. 1a). In particular, three participants reported rosacea, including facial redness and mild skin rash; one experienced adverse intestinal reactions. All four participants who dropped out experienced these adverse effects after two to three months of Collagen Activator intake, suggesting a low likelihood of an acute effect of Collagen Activator towards these events and, in general, that the Collagen Activator was safe and well-tolerated. Of the 66 participants confirmed as eligible, 37 completed the monthly questionnaires. After three months, 58 participants underwent the three skin analyses, and 45 completed the biological age test at six months (Fig. 1a)13. The 58 participants were spread across a range of 35–68 years of age. The mean age of participants was 46.91 years ± 8.06 (95% CI: 44.80–46.48) (Fig. 1b). The cohort consisted of 39 females (67.2%) and 19 males (32.8%) (Fig. 1c). The demographic and baseline characteristics of the participants were assessed using the questionnaire. The mean body mass index (BMI) was 22.28 kg/m2, with a standard deviation of 2.79 kg/m2, indicating a relatively homogeneous distribution of BMI values across the cohort (Table 2). The majority of participants (88%) were White/Caucasians (Table 2). Furthermore, 83 % of participants hold a university degree, 69% never smoked, and only 4 participants occasionally smoked, the rest all quit smoking, one-third did not drink any alcohol, and two-thirds reported either 1-2 or 3-4 drinks per week, 66% reported taking supplements, almost half of the participants exercise 3-4 times a week with 88% moderate to lightly intensity and only 2 participants reported to be sedentary (Table 2). Regarding the adherence of Collagen activator consumption. 86% of participants reported daily Collagen activator intake in the first month, which decreased to 52% at month six (P = 0.028, Fig. 1d). Taken together, our cohort with a mean age in the mid-forties is young, given that this is the start of age-related phenotypes in humans. Furthermore, our cohort is exceptionally well-educated and has healthy lifestyles.
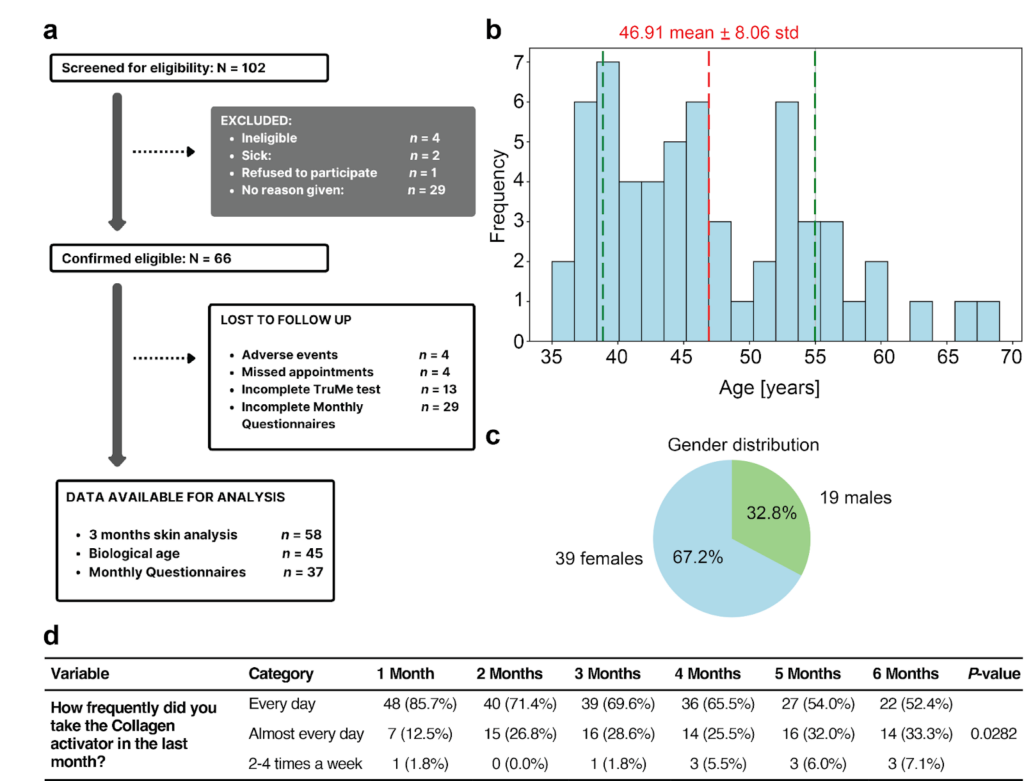
Fig.1: Clinical trial flowchart and age and sex distribution.
Table 2: Demographic and baseline characteristics.
| Variable | Category | Value |
| BMI | 22.28 ± 2.79 | |
| Ethnicity | White | 87.9% (n = 51) |
| Hispanic or Latino | 5.2% (n = 3) | |
| Asian | 3.4% (n = 2) | |
| Black or African American | 1.7% (n = 1) | |
| Not Specified | 1.7% (n = 1) | |
| Education | University Degree | 82.8% (n = 48) |
| Vocational Training | 12.1% (n = 7) | |
| Secondary School or Less | 3.4% (n = 2) | |
| Not Specified | 1.7% (n = 1) | |
| Dietary Pattern | Flexitarian | 31.0% (n = 18) |
| Omnivore | 41.4% (n = 24) | |
| Other | 15.5% (n = 9) | |
| Pescatarian | 5.2% (n = 3) | |
| Vegan | 1.7% (n = 1) | |
| Vegetarian | 3.4% (n = 2) | |
| Supplement Use | No | 32.8% (n = 19) |
| Yes | 65.5% (n = 38) | |
| Alcohol Consumption | 1-2 drinks | 31.0% (n = 18) |
| 3-4 drinks | 31.0% (n = 18) | |
| 5-8 drinks | 6.9% (n = 4) | |
| None | 29.3% (n = 17) | |
| Smoking Status | No, never | 69.0% (n = 40) |
| No, I quit | 22.4% (n = 13) | |
| Yes, occasionally | 6.9% (n = 4) | |
| Exercise Frequency | 1-2 times a week | 36.2% (n = 21) |
| 3-4 times a week | 44.8% (n = 26) | |
| 5-6 times a week | 8.6% (n = 5) | |
| Daily | 1.7% (n = 1) | |
| Less than once a week | 6.9% (n = 4) | |
| Physical Activity Level | Extremely active | 1.7% (n = 1) |
| Lightly active | 37.9% (n = 22) | |
| Moderately active | 50.0% (n = 29) | |
| Sedentary | 3.4% (n = 2) | |
| Very active | 5.2% (n = 3) |
Collagen Activator improves skin texture, hydration, and elasticity
At time point 0, i.e., baseline, participants completed a questionnaire, a skin scan of the cheek, forearm, and upper arm via Visia scan and Cutometer and Corneometer analysis at Hautwek Clinic in Zurich, and provided saliva for an epigenetic age test (Fig. 2 and Fig. 3a). Participants filled out the questionnaire every month, and during months one and three, they visited Hautwerk Zurich for skin analysis. Participants completed the questionnaire at month six and provided a saliva sample to estimate their biological age (Fig. 3a).

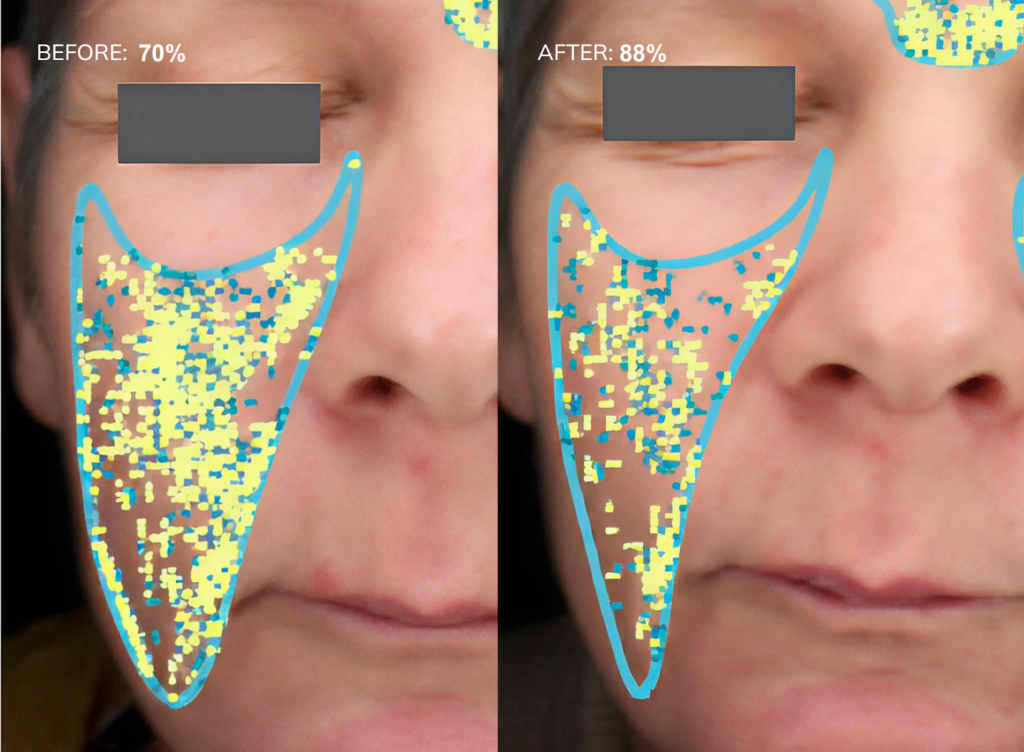
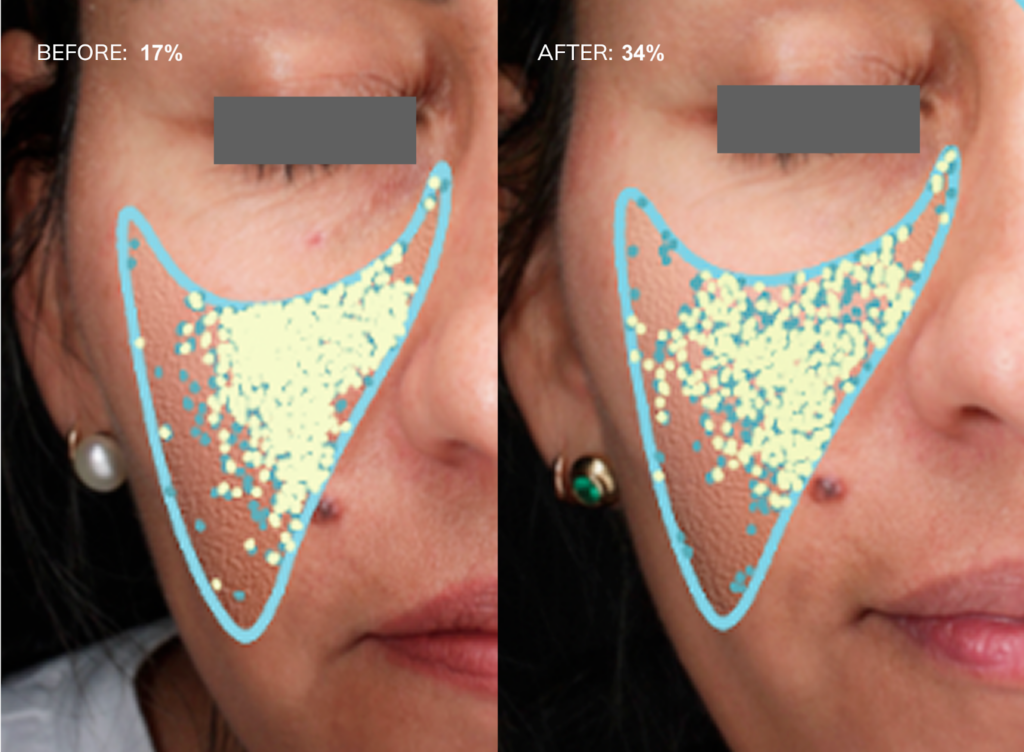
Fig.2: Examples of texture improvements via the Visia analysis, before and after 30 days of Collagen Activator intake
We found that supplementing Collagen Activator improved skin texture after one month and improved it for the rest of the study. The skin texture score increased significantly from baseline (59.93 %, 95% CI: 52.98 – 66.88 %) to the first visit (64.76 %, 95% CI: 58.14 – 71.38 %, P = 0.0435) and continued to increase until the final visit at month three (65.05 %, 95% CI: 58.35 – 71.76 %, P = 0.0116) (Fig.2 and Fig.3b).
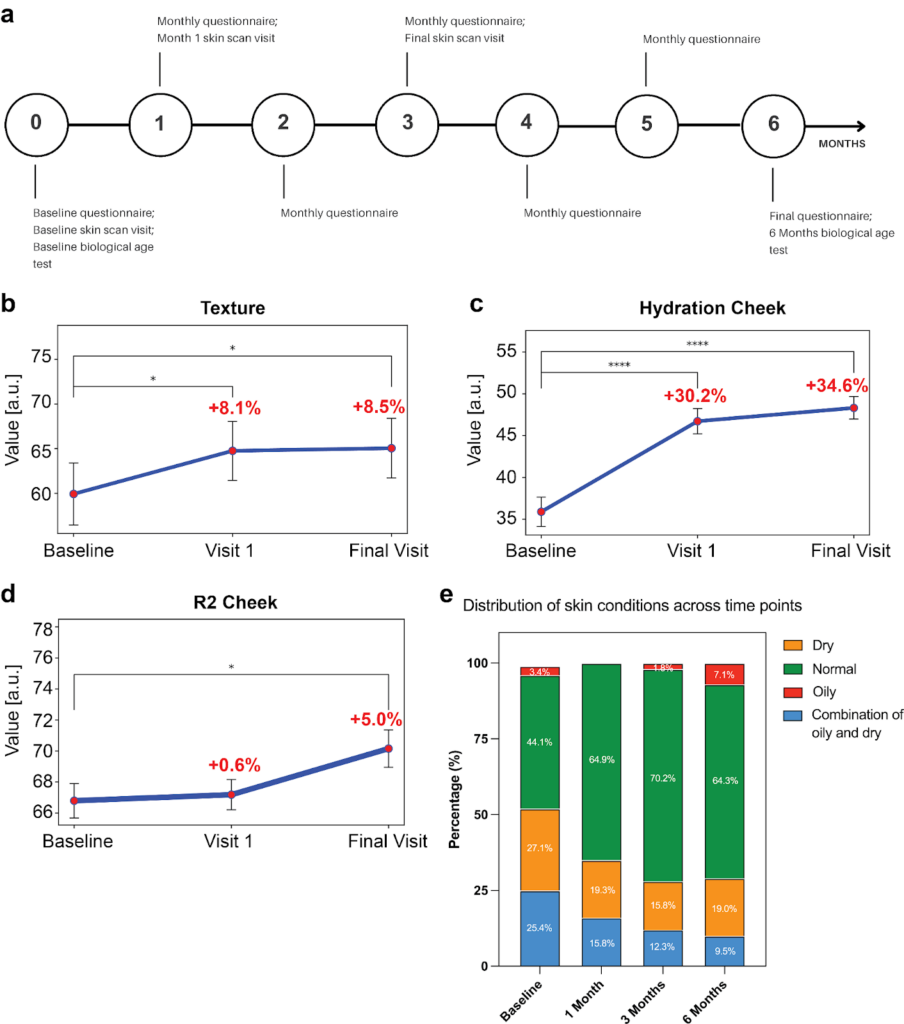
Fig.3: Changes in skin parameters over time upon Collagen Activator intake.
A representative image of the Visia analysis is shown in Figure 2, where a lower percentage indicates a less favourable skin condition, while a higher percentage reflects an improvement. In this case, the observed change from 39% to 80% signifies an improvement in skin texture – a 100% improvement – highlighting the visible efficacy of the intervention over the test period. This enhancement is also visibly evident through the reduced number of yellow dots after 30 days.
Not surprisingly, for the short duration of 3 months of the study, wrinkle severity showed a downward trend from baseline (34.07 %, 95% CI: 26.80 – 41.34 %) to the final visit (30.00 %, 95% CI: 23.80 – 36.20 %). However, this change did not reach statistical significance (P = 0.2640).
Similarly to texture, after one month of Collagen Activator intake, skin hydration improved. We measured cheek skin hydration in relative corneometer units (AU) and observed significant improvements throughout the study, increasing from a very dry baseline level (35.89 AU, 95% CI: 32.40 – 39.39 AU) to a sufficiently moistened state by the first visit (46.72 AU, 95% CI: 43.71 – 49.74 AU, P < 0.0001) and further improving by the final visit (48.32 AU, 95% CI: 45.63 – 51.01 AU, P < 0.0001) (Fig. 3c). Furthermore, forearm hydration showed significant improvement from baseline (41.96 AU, 95% CI: 39.18 – 44.74 AU) to the first visit (45.88 AU, 95% CI: 43.07 – 48.68 AU, P = 0.0046). However, this improvement was not maintained at the final visit (44.62 AU, 95% CI: 42.24 – 47.01 AU), with no significant difference observed between the baseline and final visit (P = 0.0792). Remarkably, hydration of the upper arm improved significantly over time, increasing from baseline (36.02 AU, 95% CI: 33.46 – 38.58 AU) to the first visit (42.39 AU, 95% CI: 39.57 – 45.20 AU, P = 0.0001) and remaining elevated at the final visit (40.91 AU, 95% CI: 38.44 – 43.38 AU, P = 0.0059). This suggests an improvement in skin hydration in multiple body parts.
Moreover, we also observed improvements in skin elasticity, as measured by the R2 viscoelasticity index on the cheek, which significantly improved throughout the study. The R2 index increased from the baseline (66.79%, 95% CI: 64.58 – 69.01%) to the final visit (70.16%, 95% CI: 67.75 – 72.56%, P = 0.0401), indicating enhanced skin elasticity (Fig. 3d).
Notably, we also observed a few sex-specific improvements upon Collagen Activator intake. For instance, significant changes were observed for brown spots within females between Baseline vs. Visit 1 (P = 0.0218) and Visit 1 vs. Final Visit (P = 0.0084). For red areas, males exhibited a significant reduction in red areas between Baseline vs. Final Visit (P=0.0189).
Lastly, participants reported self-perceived improvements in their skin condition up to six months (Fig. 3e). Taken together, oral supplementation of Collagen Activator improved skin texture, hydration, and elasticity within one to three months.
Collagen Activator reduces biological age
At six months, 45 participants (33 females, 12 males) completed the saliva epigenetic test to compare to the beginning (time point 0) before taking the Collagen Activator (Fig. 4a). At baseline, the mean chronological age with a standard deviation of 47.46 ± 8.33 was comparable to the mean biological age of 47.07 ± 7.21 years (mean difference = -0.39 years, 95% CI -1.80 to 1.03; P = 0.58), indicating that the participants, on average, were slightly but not statistically significant younger than expected at baseline. The linear regression coefficient was 0.715 (95% CI: 0.565-0.865, P < 0.001), displaying an adjusted R-squared of 0.682. The Pearson correlation was 0.826 (p<0.001) (95% CI: 0.703-0.901) (Fig. 4a).
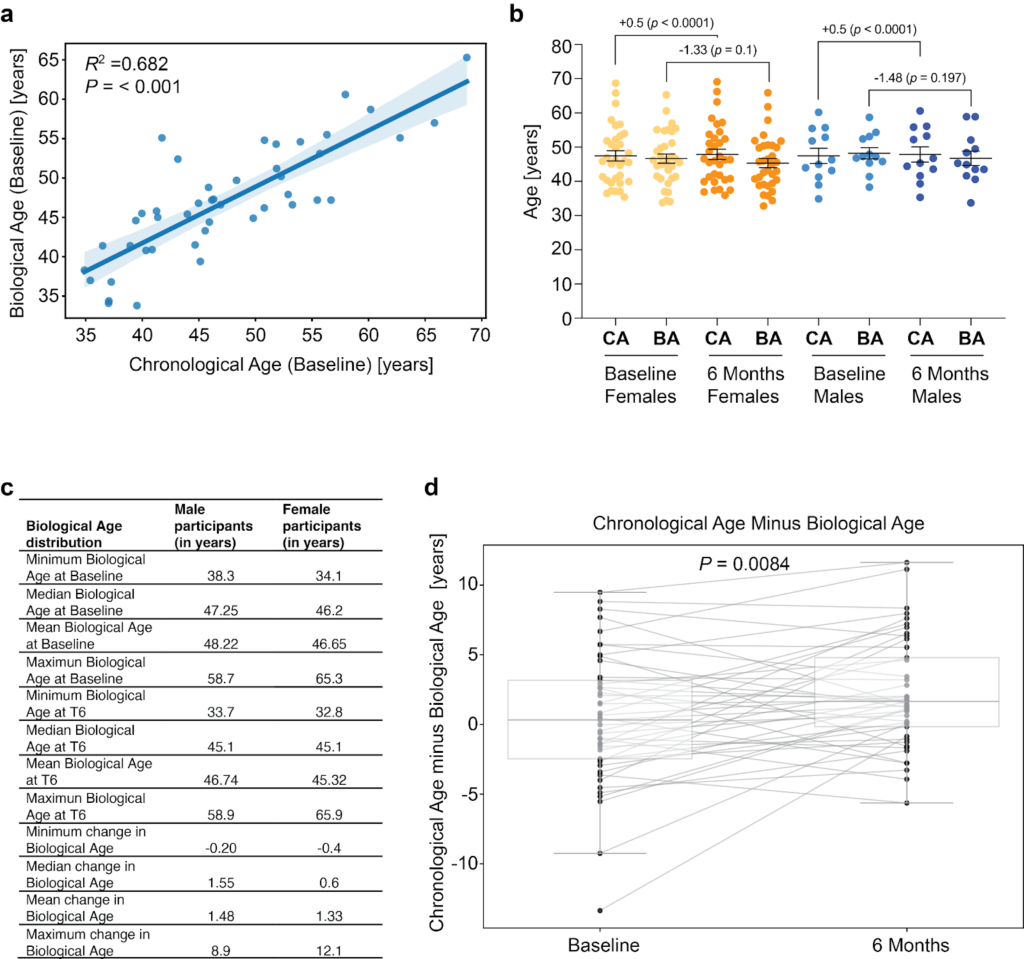
Fig.4: Improvements in biological age upon Collagen Activator supplementation.
As the natural progression of time, the chronological age increased by 0.5 years from the mean chronological age of 47.46 ± 8.33 years at baseline to 47.88 ± 8.33 years at six months. By contrast, the mean biological age at baseline was 47.07 ± 7.21 years, while the mean biological age at six months was 45.70 ± 7.57 years, demonstrating a statistically significant reduction (P = 0.040, paired t-test, two-tailed). When stratified by gender, the average decrease in biological age was similar between females (−1.33 years) and males (−1.48 years), with no statistically significant differences in the extent of biological age reduction between genders observed (Fig. 4b,c). The maximum decrease in biological age observed after 6 months of collagen supplementation was 8.9 years for males and 12.1 years for females (Fig. 4c).
To determine individual changes over six months of Collagen Activator supplementation, we plotted each participant’s chronological age minus their biological age at baseline and six months (Fig. 4d). A negative value indicates that the individual’s biological age exceeds their chronological age (i.e., ‘older’ biologically), whereas a positive value indicates the biological age is younger than their chronological age. For example, one participant had a baseline difference of -13.36 years, meaning their biological age was approximately 13 years older than their chronological age. After six months, this difference improved to -1.26, suggesting they ‘gained back’ about 12 years in six months. In contrast, another participant showed a +8.82 at baseline (i.e., their biological age was 8.82 years younger than their chronological age), which changed only slightly to +7.92 by the end of the study—indicating no benefit from supplementation on this individual’s biological age.
This might suggest that individuals with a low biological age at baseline might not benefit as much as individuals with a high biological age at baseline. To test this idea, we compared the 25 and 75 percentiles, i.e., low vs high biological age at baseline, and their change in biological age after six months of Collagen Activator supplementation. We found that individuals starting with a higher biological age than their chronological age showed the most substantial improvement in reducing their biological age after six months, with a mean of 6.3 ± 1.53 years (Fig. 5a).
Fig.5: Effects of initial biological age and lifestyle behavior on Collagen Activator’s improvements on biological age.
Next, we wondered what else could influence the improvement from Collagen Activator supplementation. The frequency of Collagen Activator use (daily vs. every other day), exercise frequency, physical activity levels, sleep quality, and education level all showed no significant effects. Although 66% of participants use other supplements, we did not find any significant difference in the biological age of participants who had taken other supplements vs. non-supplement users (Fig. 5b), suggesting that the improvements observed with the Collagen Activator are independent or synergistic to other supplements. By contrast, individuals who consumed more than one drink per week had fewer improvements with the Collagen Activator than individuals who did not drink any alcohol (Fig. 5c), suggesting a less favorable interaction between the Collagen Activator and alcohol consumption. Thus, after 6 months of using the Collagen Activator in this clinical study, we observed, on average, a 17-month reduction in biological age.
Conclusion & Implications
In this human observational study, we showed that Colgevity™, our novel and patented ratio of three amino acids, 3 glycine – 1 proline – 1 hydroxyproline, along with additional ingredients such as alpha-ketoglutarate, astaxanthin, and vitamin C, is sufficient to counteract the age-related gradual decline of collagen2. Only administering this amino acid blend already promotes health in preclinical models. Our further developed ingredient blend, showed improvements in skin features and reduced biological age by 1.33 years in women and 1.48 years in men. These findings reinforce the role of a synergy-optimised, precursor-based collagen supplementation in skin longevity. The observed benefits highlight the potential of integrating Collagen Activator into holistic longevity strategies as well as holistic skin health protocols and treatments
Material and Methods
Collagen Activator formulation
One sachet of Collagen Activator contains active ingredients: patented 8400mg Colgevity™ (5000 mg L-Glycine (Xi’an SLT Biotech Co., Ltd., Chemical synthesis), 1700 mgL-Proline (Xi’an SLT Biotech Co., Ltd., Corn fermentation), 1700 mg L-Hydroxyproline (Xi’an SLT Biotech Co., Ltd., Glucose Fermentation)), 1000 mg calcium alpha-ketoglutarate (Botanic Healthcare Pvt Ltd., Chemical synthesis), 200 mg algae powder (providing 4 mg astaxanthin; Botanic Healthcare Pvt Ltd., Chemical synthesis), and 150 mg acerola fruit juice extract (providing 48 mg vitamin C; Vidya Herbs Pvt. Ltd., Acerola fruit juice extraction).
Supporting ingredients were selected for their functional properties. Silicon dioxide was included as an anti-caking agent to ensure the powder’s free-flowing nature, improve manufacturing processes, maintain ingredient uniformity, and enhance the stability of the final product. Citric acid was added as an acidity regulator to stabilize pH, enhance the flavor, improve solubility, prevent microbial growth, and protect the product’s active ingredients, ensuring a high-quality and enjoyable final product. Steviol glycosides derived from stevia serve as a natural, calorie-free sweetener to enhance the taste, mask unpleasant flavors, and improve palatability.
For quality control, the ingredients were sourced globally, with a focus on high bioavailability and activity. Every batch underwent rigorous double-testing for purity by an independent Swiss lab.
Clinical trial design
Swissethics (Kantonale Ethikkommission Zürich) granted ethical approval for the study (BASEC ID number 2023-00953). Written informed consent was obtained from all participants after a full explanation of the study. An observational human trial was conducted, with 66 subjects recruited and informed to consume one sachet (powder) of Collagen Activator daily for 6 months. Inclusion criteria: i) Healthy adults aged above 35 years old, regardless of gender, ethnicity, or socioeconomic background; ii) Voluntarily purchasing the collagen activator (from Avea) for 6 months with the intent to take it daily iii) able to commit to 3 visits at the dermatology clinic Hautwerk in Zürich. The exclusion criteria included: i) chronic skin conditions ii) chronic medical conditions, including diabetes, cardiovascular diseases, kidney disease, liver disease, gastrointestinal disease, cancer, and autoimmune diseases; iii) pregnant and breastfeeding; iv) taking chronic medications; v) people who had any cosmetic procedures (intense pulse light, medical peelings, laser therapy, microneedling, Botox, fillers) before 3 months of the study; vi) people who had taken other collagen supplements 3 months before the first screening date; vii) Insufficient knowledge of the German or English language. Each participant underwent three skin measurements at the baseline, after 4 weeks, and after 12 weeks, respectively. Before each skin measurement, subjects were instructed to refrain from washing their face for 6 hours, use any skin care products for 12 hours, and acclimatize for 10 minutes before the visit. Self-assessment questionnaires of the subjects were collected monthly for 6 months. The biological age was measured via an epigenetic methylation clock test. Participants were asked to take the test via saliva collection at the baseline, and after 6 months, they began taking the Collagen Activator.
Skin measurements
A Visia Cam (Canfield Scientific Inc.) was used to capture in-depth insights into skin conditions. The system is based on Canfield’s RBX® technology, which utilizes cross-polarized light and UV illumination to assess reference values for spots, wrinkles, texture, pores, UV spots, red areas, and porphyrins. After image acquisition, specialized software processes the data using algorithms that detect and quantify skin parameters by identifying image patterns. The results are then compared to a comprehensive database of individuals grouped by demographic factors such as gender, age, and skin type. This database enables comparative analysis, with the software calculating a percentile for each parameter, positioning the patient’s skin condition relative to the corresponding demographic group.
A Corneometer CM 825 (Courage and Khazaka, Cologne, Germany) was used to assess skin hydration of the cheek, forearm, and upper arm. Utilizing a capacitive measurement principle, the Corneometer quantifies the moisture content of the stratum corneum by detecting changes in capacitance that occur due to water’s dielectric properties. Three measurements were performed for each location, and the values were averaged.
A Cutometer MPA 580 (Courage and Khazaka) was used to assess skin elasticity of the cheek, forearm, and upper arm, using a 2mm probe in Mode 1. The device operates based on the suction method, where negative pressure is applied to the skin, and the resulting deformation is measured to evaluate the biomechanical properties of the skin. For each measurement, a target pressure of 450 mBar was applied, with an on-time of 3 seconds and an off-time of 3 seconds, allowing the skin to recover between cycles. Five repetitions were performed at each measurement site to ensure reliability and consistency in the results. The R2 value, representing gross elasticity, assessed the skin’s overall elasticity. This parameter reflects the skin’s ability to return to its original shape after deformation, combining immediate and delayed elastic recovery.
Measurements with both the Corneometer and the Cutometer were conducted on three standardized areas: on the left cheek, at a point 5 cm from the mouth corner along the line connecting the mouth corner to the tragus, at the midpoint of the right volar forearm, and the midpoint of the medial side of the right upper arm.
The measurements were taken under controlled environmental conditions of 22°C and 45% humidity, with no daylight or windows in the room to ensure consistent conditions. Additionally, participants were instructed to refrain from contact with water and the use of cosmetics before the measurements to eliminate any external factors that could influence the results.
DNA sample collection and DNA methylation calculation
Saliva samples were taken twice: at the baseline, they were collected at Hautwerk dermatology clinic under the doctor’s supervision. After 6 months, participants collected them at home using commercially available TruMe sampling kits. After collection, the samples were shipped to TruMe Labs for analysis. DNA methylation and DNAm age have been calculated, as previously reported by Demidenko et al., 2021.
Self-reported questionnaires
Online questionnaires were sent monthly to assess product intake frequency and overall health and skin conditions.
Statistical analysis
Shapiro–Wilk test was used to assess data normality. Repeated measures ANOVA or the Friedman test was applied to evaluate changes over time. For comparisons, paired t‑tests (or Wilcoxon signed‑rank tests) were performed for matched data, and unpaired t‑tests (or Mann–Whitney U tests) were performed for independent samples. Linear regression and Pearson correlation examined associations between baseline chronological and biological age. A chi-square test was employed to evaluate associations between categorical variables. All analyses were conducted in GraphPad Prism and R, and statistical significance was set at p < 0.05.
References
- Reilly DM, Lozano J. Skin collagen through the lifestages: importance for skin health and beauty. Plast Aesthet Res. 2021;8:2. http://dx.doi.org/10.20517/2347-9264.2020.153
- Dakhovnik, A., Mantovani, M., Knufinke, M. et al. A collagen amino acid composition supplementation reduces biological age in humans and increases health and lifespan in vivo. npj Aging 11, 91 (2025). https://doi.org/10.1038/s41514-025-00280-7
- Tarnutzer, K., Siva Sankar, D., Dengjel, J., & Ewald, C. Y. (2023). Collagen constitutes about 12% in females and 17% in males of the total protein in mice. Scientific Reports, 13(1), 4490. https://doi.org/10.1038/s41598-023-31566-z
- Harkness, M. L. R., Harkness, R. D. & James, D. W. The effect of a protein‐free diet on the collagen content of mice. J. Physiol. 144, 307–313 (1958)
- Quan, T. (2023). Molecular insights of human skin epidermal and dermal aging. Journal of Dermatological Science, 112(2), 48–53. https://doi.org/10.1016/j.jdermsci.2023.08.006
- Tobin, D. J. (2017). Introduction to skin aging. Journal of Tissue Viability, 26(1), 37–46. https://doi.org/10.1016/j.jtv.2016.03.002
- Shin, J.-W., Kwon, S.-H., Choi, J.-Y., Na, J.-I., Huh, C.-H., Choi, H.-R., & Park, K.-C. (2019). Molecular mechanisms of dermal aging and antiaging approaches. International Journal of Molecular Sciences, 20(9), 2126. https://doi.org/10.3390/ijms20092126
- Straits Research, S. R. Collagen Market Size, Share & Trends Analysis Report By Source (Bovine, Porcine, Poultry, Marine, Others), By Product (Gelatin, Hydrolyzed Collagen, Native Collagen, Others), By Application (Food and Beverages, Healthcare, Cosmetics, Others) and By Region (North America, Europe, APAC, Middle East and Africa, LATAM). Straits Research https://straitsresearch.com/report/collagen-market.
- Gibson, R., Krug, L., Ramsey, D. L., Safaei, A., & Aspley, S. (2024). Beneficial effects of multi-micronutrient supplementation with collagen peptides on global wrinkles, skin elasticity and appearance in healthy female subjects. Dermatology and Therapy, 14(6), 1599–1614. https://doi.org/10.1007/s13555-024-01184-2
- Danessa, G., Notario, D., & Regina, R. (2025). Effects of collagen-based supplements on skin’s hydration and elasticity: A systematic review and meta-analysis. Indian Journal of Dermatology, Venereology and Leprology, 91(6), 730–740. https://doi.org/10.25259/IJDVL_1165_2023
- Jadach, B., Mielcarek, Z., & Osmałek, T. (2024). Use of collagen in cosmetic products. Current Issues in Molecular Biology, 46(3), 2043–2070. https://doi.org/10.3390/cimb46030132
- Al-Atif, H. (2022). Collagen supplements for aging and wrinkles: A paradigm shift in the fields of dermatology and cosmetics. Dermatology Practical & Conceptual, 12(1), e2022018. https://doi.org/10.5826/dpc.1201a18
- Demidenko, O., Barardo, D., Budovskii, V., Finnemore, R., Palmer, F. R., Kennedy, B. K., & Budovskaya, Y. V. (2021). Rejuvant®, a potential life-extending compound formulation with alpha-ketoglutarate and vitamins, conferred an average 8 year reduction in biological aging, after an average of 7 months of use, in the TruAge DNA methylation test. Aging, 13(22), 24485–24499. https://doi.org/10.18632/aging.203736












