Abstract
Background: Sleep plays a central role in human health, and disturbances in sleep continuity or recovery are closely linked to impaired metabolic function, immune response, and emotional resilience. Traditional pharmacological treatments often carry risks of side effects or dependency, highlighting the need for natural alternatives. Sereniser is a novel formulation combining clinically studied botanical extracts and key nutrients designed to support relaxation, stabilize circadian rhythms, and promote restorative sleep by targeting GABAergic and serotonergic signaling pathways.
Objective: To evaluate the effects of AVEA Sereniser on sleep quality and recovery through two independent pilot studies (AVEA collaborated with PRUVN (USA) and Quest Vitality (Germany)), using subjective sleep and mood questionnaires, objective biomarker tracking via Oura Ring, and open-ended participant feedback.
Results: Both studies reported consistent improvements in sleep continuity, morning recovery, and subjective vitality, with no side effects and high acceptance of Sereniser.he formulation’s ingredients—such as Safr’Inside™ (saffron extract), Holixer™ (holy basil), Serezin™ (ginger and boswellia), L-Theanine, Magnesium Bisglycinate, and Vitamin B6—work synergistically to reduce cortisol levels, enhance parasympathetic tone, and support endogenous sleep mechanisms without sedation or melatonin supplementation.
Conclusion: Sereniser offers a promising natural, non-pharmacological approach to improving mild to moderate sleep disturbances, particularly relevant for populations affected by stress, hormonal changes, or aging. These preliminary findings support further research into its efficacy for enhancing sleep quality and recovery using both subjective and objective measures.
Keywords: Sleep and recovery, GABA, Parasympathetic activity, alpha brainwaves
In this study
Introduction
Sleep plays a central role in human health, and disturbances in sleep continuity or recovery are closely linked to impaired metabolic function1, immune response2, and emotional resilience3. Sleep architecture, continuity and restorative recovery are fundamental to long‑term health yet adults frequently experience disruptions related to stress, hormonal shifts and ageing. Sleep disturbances, whether acute or chronic, trigger a cascade of pathological responses across multiple physiological systems4. In the short term, insufficient or disrupted sleep impairs executive function, reduces attention span and can increase the risk of accidental injury5. Chronic sleep disturbance is a primary driver for metabolic and cardiovascular disorders, including glucose intolerance, increased systemic inflammation, a heightened risk of hypertension and suppressed immune signaling6,7.
Chronic sleep challenges affect 10–30% of adults globally8 and are increasingly tied to ageing, stress, and lifestyle imbalances. These challenges are exacerbated by modern ‘circadian mismatch’ factors such as excessive blue light exposure, chronic stress and regular consumption of stimulants9. In addition, specific life-stage transitions-particularly the peri- and postmenopausal phases in women—introduce distinct physiological challenges, including hormonal fluctuations and increased musculoskeletal discomfort, both of which can further destabilize sleep architecture10,11. Traditional pharmaceutical interventions often come with side effects or dependency risks. In this context, there is growing interest in non-pharmacological interventions that support endogenous sleep regulation rather than overriding it through sedative mechanisms.
Background: Sleep and Recovery
Sleep is regulated by the homeostatic sleep drive (Process S) and the circadian rhythm (Process C)12. Process S represents the accumulation of sleep pressure during wakefulness, driven by molecules such as adenosine. Process C is regulated by the suprachiasmatic nucleus (SCN) of the hypothalamus, which synchronizes physiological processes to the light–dark cycle. For restorative sleep to occur, these two processes must be synchronised with neurochemical signalling pathways.
The architecture of sleep is divided into two distinct alternating stages- Non-Rapid Eye Movement (NREM) and Rapid Eve Movement (REM). During NREM sleep, particularly slow wave sleep (SWS), the brain undergoes significant metabolic clearance through the glymphatic system (a specialised waste clearing pathway in the brain), while the body engages in tissue repair and immune system modulation13. REM sleep conversely, is vital for emotional processing and consolidating procedural and spatial memory13.
The transition from wakefulness to sleep is mediated by the inhibition of the brain’s arousal systems, including noradrenergic, histaminergic, and serotonergic pathways12. This process is orchestrated by the ventrolateral preoptic nucleus (VLPO), which releases gamma-aminobutyric acid (GABA), the brain’s primary inhibitory neurotransmitter, to suppress wake-promoting signals. Simultaneously, melatonin secretion increases in response to darkness, signaling the onset of the biological night12. Sleep interventions must target multiple nodes within this regulatory framework.
Mechanistic Positioning of the Sereniser Formulation
The AVEA Sereniser formulation is designed to target key biological pathways, characterised by a synergy of clinically‑studied botanical extracts (Safr’Inside™, Holixer™, Serezin™) and key nutrients (L‑Theanine, Magnesium Bisglycinate, Vitamin B6) with the objective of supporting endogenous sleep mechanisms without sedation or melatonin supplementation. The formulation’s mechanism of action, targeting GABAergic and serotonergic signalling, reducing cortisol levels, and enhancing parasympathetic tone, is biologically aligned with the needs of individuals experiencing sleep disruption due to stress, hormonal transition, or ageing.
Safr’Inside™, a standardised saffron extract, is shown to support serotonin‑melatonin conversion and cortisol reduction; Holixer™ (holy basil extract) supports parasympathetic activity and cortisol regulation; Serezin™ (ginger + boswellia) addresses nocturnal discomfort and supports deep, uninterrupted sleep. Furthermore, L‑Theanine promotes alpha brain wave activity and mental calm, Magnesium Bisglycinate aids GABA signalling and muscle relaxation, and Vitamin B6 acts as a cofactor in GABA and serotonin synthesis. Therefore, Sereniser offers a natural, non-pharmacological solution formulated with plant-based and evidence-backed ingredients. Its mechanism targets GABAergic and serotonergic signalling, both of which regulate anxiety, relaxation, and sleep architecture. This makes Sereniser particularly relevant for populations experiencing mild to moderate sleep disturbances, including peri- and postmenopausal individuals.
Table 1: Key Active Ingredients and Mechanistic Roles
| Ingredient | Primary mechanistic role | Expected clinical outcomes in Aging | Citations |
| Safr’Inside™ | Supports serotonin-melatonin conversion and modulates the HPA axis to delay cortisol peaks. | Improved sleep efficiency and significantly reduced sleep latency in older adults (ages 55–85);Reduction in perceived stress and moderate insomnia symptoms. | |
| Holixer™ (holy basil extract) | Reduces chronic cortisol output; enhances parasympathetic tone and stabilizes sleep microarchitecture (CAP A1 activity) | Improvement in insomnia scores; reduced nighttime awakenings and lower cortisol levels (a marker of long-term stress). | |
| Serezin™ (ginger + boswellia) | Dual inhibition of COX-2 and 5-LOX inflammatory pathways; reduces pro-inflammatory cytokines like TNF-$\alpha$ to address musculoskeletal discomfort. | Relief from nighttime and daytime aches; Improvement in sleep quality and restorative sleep; supports deep, uninterrupted sleep; improved morning alertness and mood. | |
| L‑Theanine (green tea extract) | Induces alpha brainwave activity (relaxed alertness); acts as a GABA agonist and weak glutamate antagonist to block neuronal overexcitation | Reduced “racing thoughts” before bed; significantly higher sleep percentage and efficiency; enhanced feelings of recovery and refreshment upon waking. | |
| Magnesium Bisglycinate | GABA agonist and NMDA antagonist; essential cofactor for melatonin synthesis; glycine component promotes thermoregulation (lowering core body temperature). | Significant increase in total sleep time and efficiency in elderly populations; reduction in insomnia severity (ISI) and nighttime muscle tension or cramps. | |
| Vitamin B6 | Rate-limiting cofactor for enzymes GAD (GABA synthesis) and AADC (serotonin synthesis); essential for amino acid metabolism. | Correlation with lower insomnia scores and higher sleep quality; reduction in anxiety symptoms; synergistic relief of Restless Leg Syndrome (RLS) when paired with magnesium. |
In this observational human study, we aimed at exploring preliminary clinical data collected from two independently conducted pilot studies that investigated the AVEA Sereniser’s effect profile on sleep using a wearable finger ring tracker (Oura ring)5 and self-reported questionnaires.
Results & Discussion
Self-reported sleep quality
A structured questionnaire-based evaluation conducted by QuestVitality assessed subjective changes in sleep and recovery among six adult participants (age range: 30–65 years; balanced gender distribution) over a 30-day supplementation period with Sereniser. Participants maintained high adherence (≥90%), and no relevant adverse events were reported.
Over 30 days, participants consistently reported improvements in multiple sleep dimensions. Two-thirds of the cohort noted easier sleep initiation, while five of six described fewer nocturnal awakenings, indicating enhanced sleep continuity. A similar proportion reported feeling more rested upon waking, and four participants also experienced increased daytime energy and alertness—suggesting improved overall sleep quality and recovery (Fig. 1).
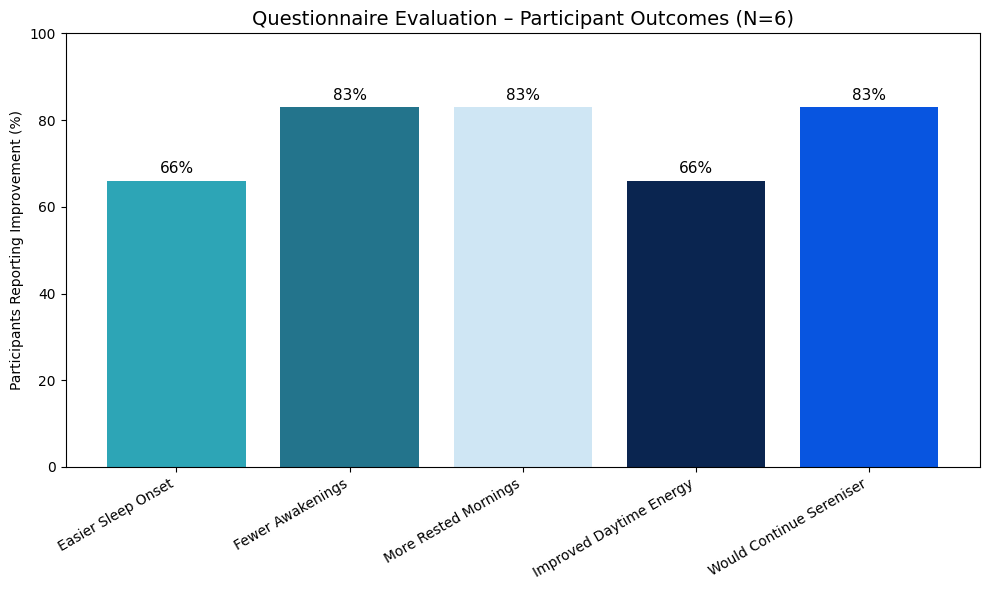
Figure 1. Quest Vitality questionnaire evaluation. Participant outcomes of subjective sleep quality parameters.
In the PRUVN pilot study, participants reported improvements in the self-reported questionnaires in three dimensions related to sleep and daily functioning after 30 days of Sereniser supplementation. On average, sleep quality improved by 78%, fatigue levels decreased by 61%, and mood improved by 48% compared to baseline (Fig 2.).
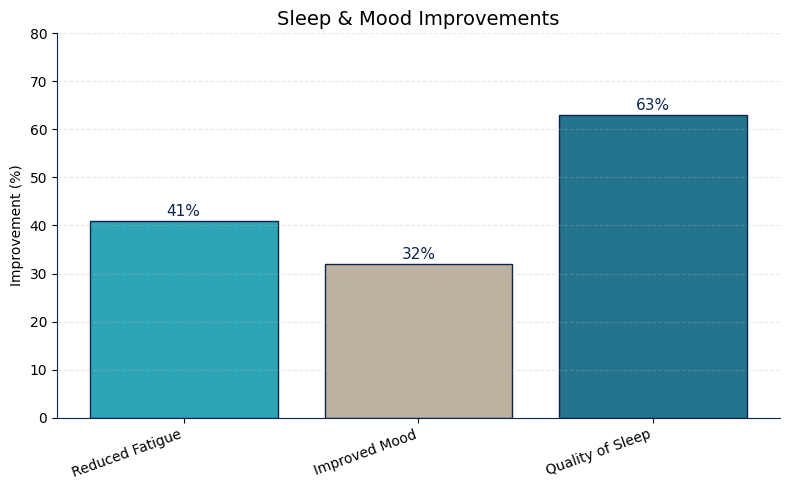
Figure 2. PRUVN questionnaire evaluation. Participant outcomes of subjective sleep quality parameters.
Sleep biomarkers via wearable tracker
Objective sleep data were collected via Oura Ring monitoring in two independent, observational pilot studies conducted by QuestVitality and PRUVN. Participants completed a 15-day baseline phase followed by a 30-day intervention phase with daily Sereniser supplementation. The QuestVitality study involved eight peri- and postmenopausal women aged 52–60, while the PRUVN study included eight participants (mixed sex) over 40 years old, selected based on self-reported sleep complaints and prior Oura Ring use. In both cohorts, consistent trends toward improved sleep physiology were observed.
In the Quest Vitality study, The most notable effects were:
- Sleep Efficiency: Improved in 3 out of 4 participants (average +3–5%), indicating more consolidated and restorative sleep (Fig. 3);
- Heart Rate Variability (HRV): Increased in all participants (+7–25%), suggesting enhanced parasympathetic activity and better overnight recovery (Fig. 3);
- Resting Heart Rate: Decreased in all cases (–3 to –5 bpm), reflecting improved cardiovascular recovery and autonomic balance (Fig. 3);
- Total Sleep Duration: Slightly increased in most participants (average +10–20 minutes), supporting deeper, more restorative sleep phases (Fig. 3);
- Sleep Latency: Decreased (–10–20%), implying faster sleep onset and reduced pre-sleep arousal (Fig. 3).
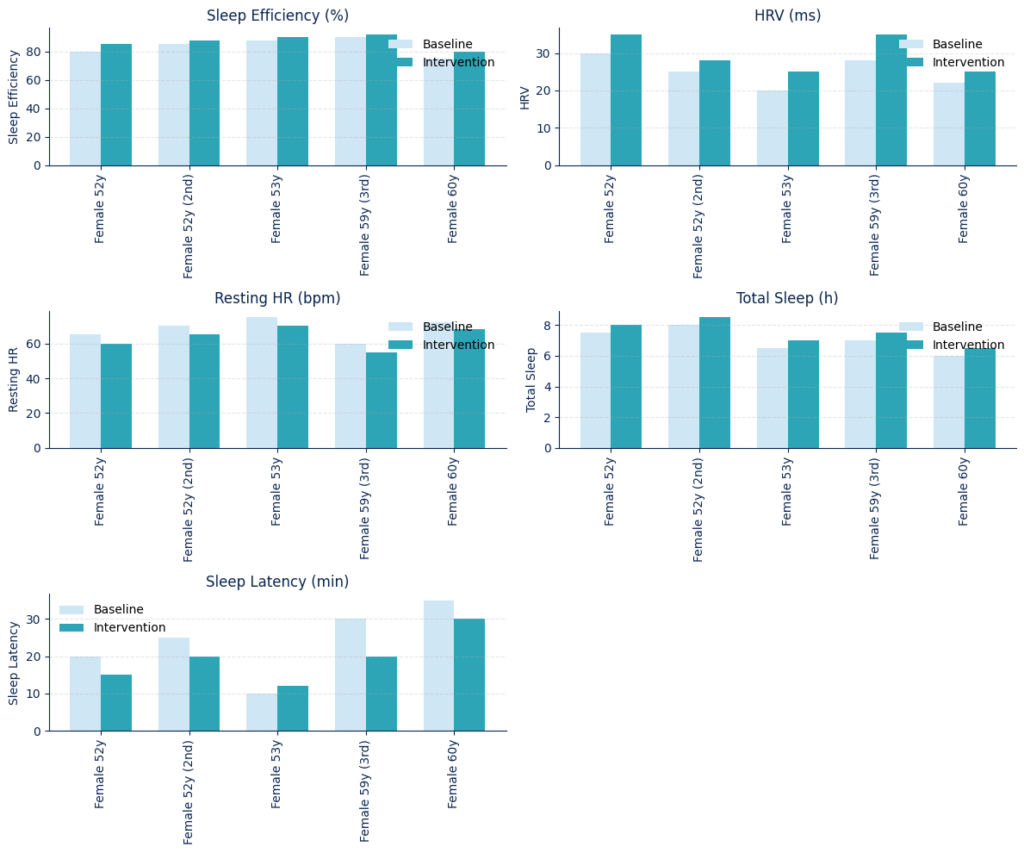
Figure 3. Quest Vitality comparative sleep analysis. Oura-ring metrics analyzed include Sleep Efficiency, Heart Rate Variability (HRV), Resting Heart Rate, Total Sleep Duration, and Sleep Latency. Data were captured via Oura Ring monitoring.
In the PRUVN pilot study, the data showed consistent improvements in key sleep metrics compared to baseline:
- REM sleep increased by up to 22% (Fig. 4);
- Deep sleep improved by up to 5% (Fig. 4);
- Total sleep duration increased by as much as 21% (Fig. 4);
- Sleep efficiency improved by up to 9%, while sleep latency was reduced by 18% (Fig. 4);
- Overall sleep score increased by up to 5% (Fig. 4).
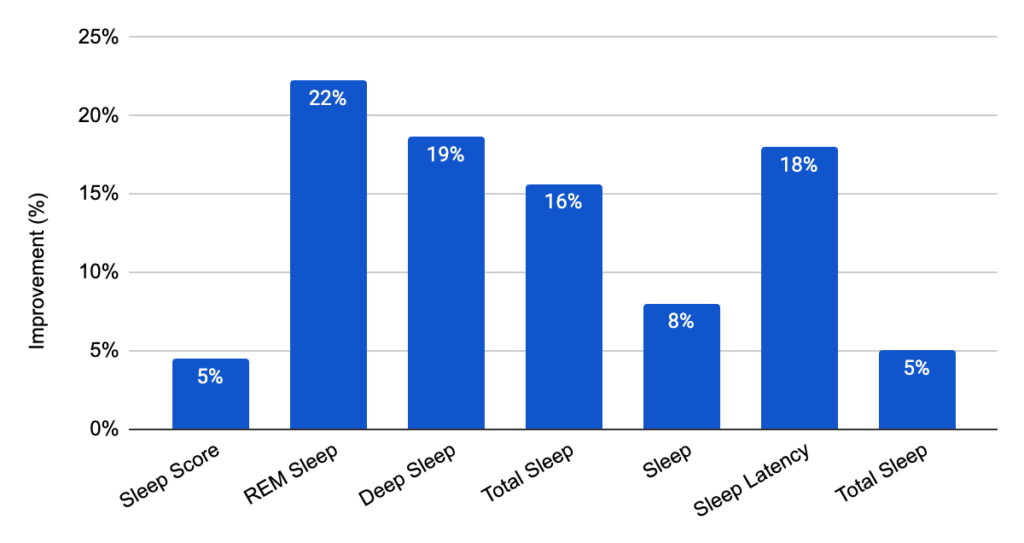
Figure 4. PRUVN objective measurements. Oura Ring data show increases in REM and deep sleep, total sleep duration, and efficiency, with reduced sleep latency compared to baseline.
These improvements suggest that Sereniser positively influenced both sleep architecture and quality, with notable gains in restorative sleep phases (REM and deep sleep).
Adverse Events Monitoring
Across both studies, no adverse events or intolerances were reported. Sereniser was well tolerated by all participants, with no incidents of discontinuation due to side effects. Overall, the safety profile of Sereniser was rated as very good, supporting its suitability for use in adults experiencing mild to moderate sleep disturbances.
Conclusion & Implications
The combined findings from these exploratory studies indicate that Sereniser may offer a safe and well-tolerated approach to improving sleep quality, continuity, and physiological recovery in adults with mild to moderate sleep disturbances. Both subjective assessments and objective biometric data showed consistent trends, notably in sleep onset, maintenance, HRV, resting heart rate and sleep efficiency. The formulation’s mechanism of action, targeting GABAergic and serotonergic signalling, reducing cortisol levels, and enhancing parasympathetic tone, is biologically aligned with the needs of individuals experiencing sleep disruption due to stress, hormonal transition, or ageing. In particular, we deliberately included a cohort of peri- and postmenopausal women, a population known to be disproportionately affected by sleep fragmentation and circadian rhythm instability. In this group, both subjective and objective data showed clear positive trends, especially in sleep consolidation and next-day recovery metrics, suggesting Sereniser may hold particular relevance for this demographic. These attributes position Sereniser as a compelling alternative to melatonin‑based interventions, particularly for individuals seeking a physiologically‑aligned solution that supports the body’s natural sleep architecture.
Although limitations include small sample sizes and lack of placebo control, the consistent directionality of outcomes across independent studies provides a strong rationale for further research.
Material and Methods
Study design
This study summarises the outcomes from two independent and non-randomised open label pilot studies conducted in collaboration with PRUVN and Quest Vitality. The studies were designed to evaluate the effectiveness of AVEA Sereniser supplementation on sleep quality, recovery, and daytime energy over an intervention period of 30 days. No placebo group was included, and participant measurements were compared pre- and post-intervention. Participants were instructed to take two capsules of Sereniser with water, every evening, 1–2 hours before bedtime. Participants cohort:
- QuestVitality study:
N = 8 adult women (aged 52–60), all in peri- or postmenopausal stage, reporting mild to moderate sleep complaints. - PRUVN study:
N = 8 adults (mixed sex, aged >40), with self-reported non-clinical sleep disturbances and access to validated biometric tracking (Oura Ring).
Participants were recruited based on inclusion criteria i) Age ≥ 40 years; ii) Not currently taking prescription sleep medications; iii) No change in medication, supplement, or lifestyle factors during study period; iiii) Consent to daily supplementation and continuous biometric tracking. All participants provided informed consent for the use of anonymised data for research and product development purposes.
Oura Ring measurements
Objective sleep and recovery metrics were collected using the Oura Ring, a medical-grade wearable device that continuously measures biometrics through photoplethysmography (PPG), a non-invasive optical method that tracks changes in blood volume to assess physiological parameters. Participants wore the Oura Ring continuously during the 30-day baseline phase and the 30-day supplementation phase. Key sleep-related metrics extracted and analysed included: i) Heart Rate Variability (HRV): Marker of autonomic nervous system balance and parasympathetic recovery; ii) Resting Heart Rate (RHR): Indicator of cardiovascular efficiency and sleep depth; iii) Sleep Efficiency: Proportion of time in bed spent asleep; iv) Sleep Latency: Time taken to transition from wakefulness to sleep; vi) Total Sleep Duration: Total minutes of sleep per night. Data were automatically synchronised with the Oura app and manually extracted for anonymised comparison between baseline and post-intervention periods.
Self-reported questionnaires
Participants completed structured pre- and post-intervention questionnaires assessing: i) Sleep onset and continuity; ii) Morning refreshment and perceived sleep quality; iii) Daytime alertness and energy; iv) Overall product satisfaction and tolerability; vi) Willingness to continue using Sereniser. Ratings were collected using Likert-type scales (1–10), complemented by open-text responses.
References
- Knutson KL, Spiegel K, Penev P, Van Cauter E. The metabolic consequences of sleep deprivation. Sleep Med Rev. 2007 Jun;11(3):163-78. doi: 10.1016/j.smrv.2007.01.002. Epub 2007 Apr 17. PMID: 17442599; PMCID: PMC1991337.
- Garbarino S, Lanteri P, Bragazzi NL, Magnavita N, Scoditti E. Role of sleep deprivation in immune-related disease risk and outcomes. Commun Biol. 2021 Nov 18;4(1):1304. doi: 10.1038/s42003-021-02825-4. PMID: 34795404; PMCID: PMC8602722.
- Cara C Tomaso, Anna B Johnson, Timothy D Nelson, The effect of sleep deprivation and restriction on mood, emotion, and emotion regulation: three meta-analyses in one, Sleep, Volume 44, Issue 6, June 2021, zsaa289, https://doi.org/10.1093/sleep/zsaa289
- Porkka-Heiskanen, T., Zitting, K.-M., & Wigren, H.-K. (2013). Sleep, its regulation and possible mechanisms of sleep disturbances. Acta Physiologica (Oxford, England), 208(4), 311–328. https://doi.org/10.1111/apha.12134
- Medic, G., Wille, M., & Hemels, M. E. (2017). Short- and long-term health consequences of sleep disruption. Nature and Science of Sleep, 9, 151–161. https://doi.org/10.2147/NSS.S134864
- Miller, M. A., & Cappuccio, F. P. (2007). Inflammation, sleep, obesity and cardiovascular disease. Current Vascular Pharmacology, 5(2), 93–102. https://doi.org/10.2174/157016107780368280
- Zhao, X., Li, Y., & Wang, T. (2025). Effects of sleep patterns on cardiovascular health. Cardiovascular Innovations and Applications, 10(1). https://doi.org/10.15212/cvia.2025.0022
- Georgiev T, Draganova A, Avramov K, Terziyski K (2025) Chronic insomnia – beyond the symptom of insufficient sleep. Folia Medica 67(3): e151493. https://doi.org/10.3897/folmed.67.e151493
- Nobre, B., Rocha, I., Morin, C. M., & Cruz, M. M. E. (2021). Insomnia and circadian misalignment: an underexplored interaction towards cardiometabolic risk. Sleep Science (Sao Paulo, Brazil), 14(1), 55–63. https://doi.org/10.5935/1984-0063.20200025
- Virtanen, I., Polo-Kantola, P., Turpeinen, U., Hämäläinen, E., & Kalleinen, N. (2023). Effect of external sleep disturbance on sleep architecture in perimenopausal and postmenopausal women. Climacteric: The Journal of the International Menopause Society, 26(2), 103–109. https://doi.org/10.1080/13697137.2022.2158727
- Toffol, E., Kalleinen, N., Himanen, S.-L., Partonen, T., Haukka, J., & Polo-Kantola, P. (2021). Nighttime melatonin secretion and sleep architecture: different associations in perimenopausal and postmenopausal women. Sleep Medicine, 81, 52–61. https://doi.org/10.1016/j.sleep.2021.02.011
- Franken, P., & Dijk, D.-J. (2024). Sleep and circadian rhythmicity as entangled processes serving homeostasis. Nature Reviews. Neuroscience, 25(1), 43–59. https://doi.org/10.1038/s41583-023-00764-z
- Chopra, A., Das, P., & Doghramji, K. (Eds.). (2020). Management of sleep disorders in psychiatry. Oxford University Press. https://doi.org/10.1093/med/9780190929671.001.0001
- Herberger, S., Aurnhammer, C., Bauerfeind, S. et al. Performance of wearable finger ring trackers for diagnostic sleep measurement in the clinical context. Sci Rep 15, 9461 (2025). https://doi.org/10.1038/s41598-025-93774-z












